This is a jump from 2D to 3D structure representation where we can visually picture how a molecule actually remains in bonding nature in reality. If you want to dive deeper into how the inside of a molecule looks like, we need to learn about molecular geometry. 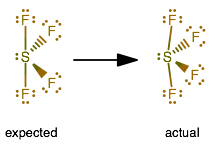
Note: This is an exception to the octet rule since xenon has more than eight electrons in this compound in its valence shell cloud. Therefore, since both the elements are in their least possible formal charge values, we have got our most suitable Lewis Structure. This is a necessary topic for chemical bonding, especially for Lewis Structure determination where we need to check the least possible formal charges of each combining atom to get the perfect diagrammatic representation.įor Xe, formal charge= 8 (valence electron number) – 0.5*4 (number of bonded electrons) – 6 (no of lone pair electrons) = 0įor each fluorine atom, formal charge= 7 – 0.5*2 – 6 = 0 If we get to assume that every electron is shared equally among atoms, we can assign a formal charge to an atom. So, we keep the valence electrons around xenon and calculate the formal charge. Now, the obvious choice is to form a double bond with the remaining two valence electrons instead of single bonds.įluorine is so electronegative it doesn’t usually form double bonds. Now, as we can see we have achieved the octet fulfillment for the three atoms inside XeF2 and single bond formation has been done.īut if you count the total valence electrons in the diagram, we can see the dots add up to 20 and not 22. eight electrons outside each atom in its outer shell) Now that we have the central atom and the total valence electron number, we will find out how the electrons are located to reach the octet configuration( i.e. If you go by the periodic table, we know that xenon is less electronegative than fluorine, hence it will take up the position of a central atom. Total number of valence electrons =8 + 7*2 = 22 Fluorine is a halogen belonging to group VII, therefore it has a valency of seven. Therefore, it has eight valence electrons. We use dots to represent outer shell electrons and lines to represent the bond type. Lewis Structure, also known as electron dot structure, is an essential model of chemical bonding where we use the valence electron concept to schematically sketch a two-dimensional figure of a given molecule. If you go through the periodic table, the groups will help us find out the valence electron number of a certain atomic element.įor example, Carbon is in group 4 (also called Group XIV), hence the valence shell consists of four electrons. Valence electrons signify the outermost shell electrons of an atom that determine its valency. The very first thing to learn when we are into chemical bonding is the concept of valence electrons. This is known as chemical bonding which is the backbone to define the internal structure and nature of a given molecular compound including the properties it exhibits( both physical and chemical).īefore we jump directly into the chemical bonding of XeF2 in this article, we would like you to learn and recapitulate certain important terminologies and concepts. This formation of molecules happens via the creation of certain bonds which hold the atoms together according to their strength. When two or more atoms come together they react and combine to form homogeneous and heterogeneous molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
